India: Health Ministry Issues Grouping Guidelines For Medical Device Applications
Last Updated: 19 April 2018
Article by Singh & Associates
Singh & Associates
On March 16, 2018, the Ministry of Health and Family Welfare (MoH&FW) issued the grouping guidelines of Medical Devices in pursuance of Rule 5 of the Medical Devices Rules (MDR), 2017. This guidance is for a person who applies for license to import or manufacture for sale or distribution of medical devices, namely –
- Application for license to import or manufacture for sale or distribution, sell, stock or offer for sale or distribution of medical device under Medical Devices Rules, 2017.
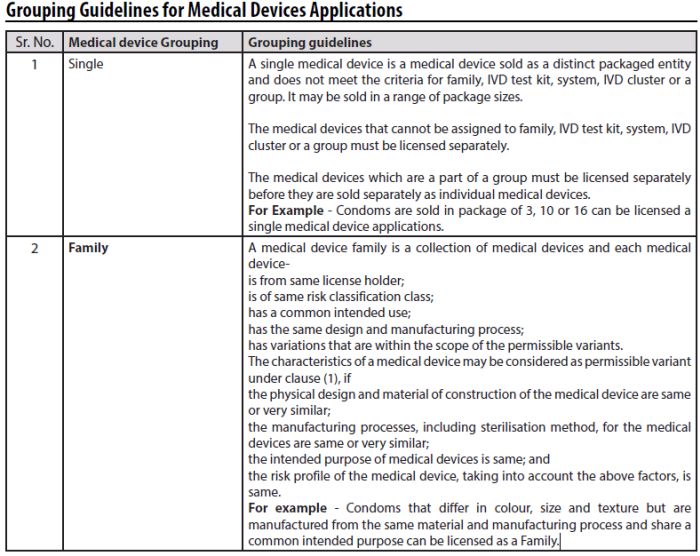
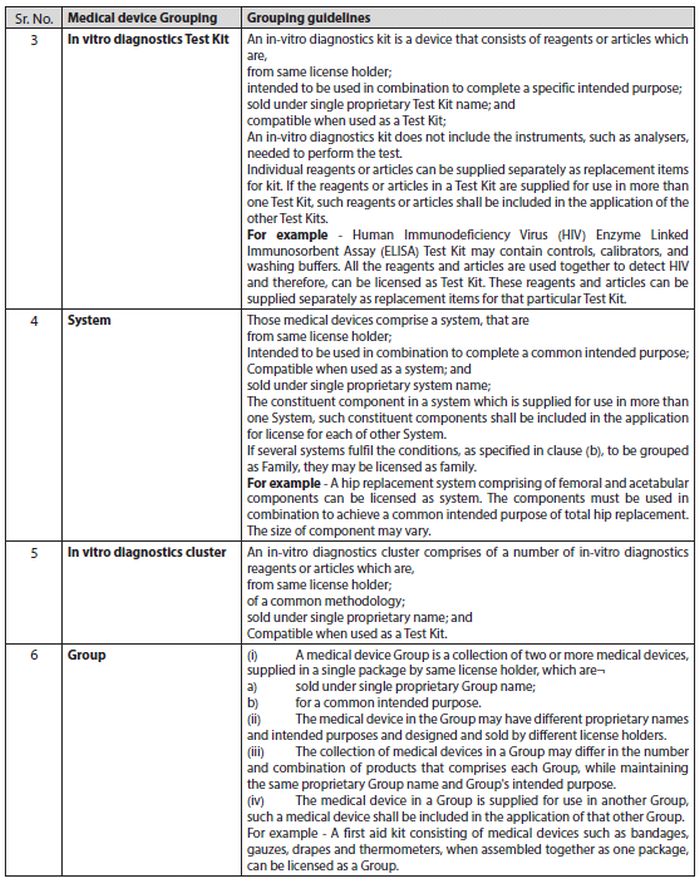
- The applicant may group medical devices having same or similar intended uses or commonality of technology and submitted in a single application. The grouping of medical devices is for the purpose of submission of single application for license to import or manufacture in the manner (Tabulated below).
The guidelines have been prepared in accordance with the Medical Devices Rules, 2017, categorizing medical devices into six brackets namely 1) Single, 2) Family, 3) In-vitro Diagnostic Test Kit, 4) System, 5) In-vitro Diagnostic Cluster, and 6) Group. An applicant is allowed to group medical devices having same or similar intended uses or commonality of technology and submit a single application for a license to manufacture or import it.
Conclusion:
The grouping guidelines provides detailing on when and how companies seeking clearance to import or manufacture medical devices can group products in a single submission. This grouping guideline supports the Medical Devices Rules, 2017.
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.